Features & Benefits
A Clinically Economic Solution
23% Average Savings per Year
With the HeRO Graft predicted by a US healthcare model for provision of dialysis access compared to catheters.5
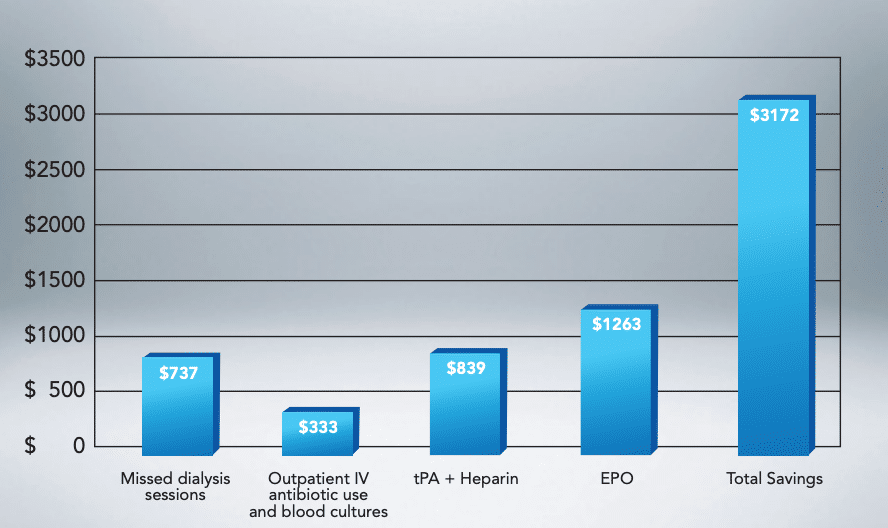
Cost Savings of Over $3,100 (per Patient/Year)
To the dialysis center when converting catheter-dependent patients to the HeRO Graft.6
Lowers the Number of Interventions
By more than 50% compared to catheters.7, 8
Reduces Device-Related Infection Compared to Catheters ^7
That can result in hospital admissions projected at $23K to $56K per stay9, 10
Impact of HeRO Graft in the Era of Dialysis Provider Bundling
REFERENCES:
1. Atrium Medical Corp. Product Catalog, Vascular Grafts, Vascular Patches and Tunnelers, Dec. 2, 2011. Product Flixene® Vascular Graft, Standard Wall. http://www.atriummed.com/EN/vascular/Documents/ US%20Grafts%20Catalog.pdf, downloaded on 08/29/2013.
2. W. L. Gore & Associates. GORE® ACUSEAL Vascular Graft Congurations. http://www.goremedical.com/acusealvg/congurations/, downloaded on 08/29/2013.
3. W. L. Gore & Associates. GORE-TEX® Standard-walled Vascular Grafts. http://www.goremedical.com/contentTypeDetail.jsp?action=contentDetail&N=8096%208239&contenttype=8327&R=1234726363369, downloaded on 08/29/2013.
4. C. R. Bard. Bard Peripheral Vascular 2013 Vascular Product List. S120082 Rev1. http://www.bardpv.com/pdfs/2013-Vascular-Product-List.pdf, downloaded on 08/29/2013.
5. Dageforde et al., JSR 2012.
6. Yost and Dinwiddie, Poster Presentation at American Society of Nephrology (ASN) Meeting 2010.
7. Katzman et al., J Vasc Surg 2009.
8. Gage et al., EJVES 2012.
9. Ramanathan et al, Infect Control Hosp Epidemiol 2007.
10. O’Grady et al, Pediatrics Nov 2002.
HeRO Graft is classified by the FDA as a vascular graft prosthesis. For the grafts permitted for use with the Adapter and Support Seal, refer to the graft manufacturer.
Instructions for Use. *When used with HeRO 1001 and HeRO 1002, similar results are expected when used with the HeRO 1000 Adapter.
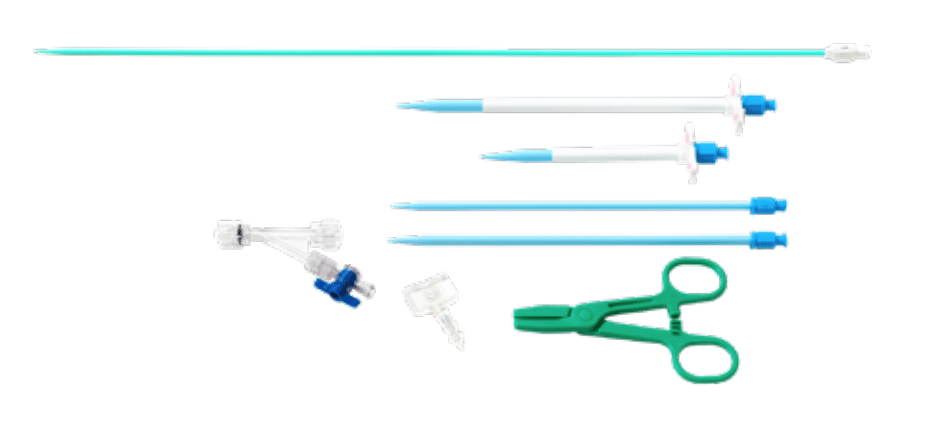
Accessory Component Kit
- 10F Delivery Stylet
- 12F and 16F Dilators
- 20F Peel Away Sheath with Dilator (Long & Short Options)
- Disposable Clamp
- Hemostasis Plug
- Y-Adapter with 1-Way Stopcock
GIVING YOU MORE GRAFT OPTIONS TO BYPASS CENTRAL VENOUS STENOSIS
The Super HeRO® Adapter and Support Seal system expands upon the clinically proven HeRO Graft by allowing surgeons to utilize other standard wall and early cannulation vascular graft options.
APPROVED EARLY CANNULATION* VASCULAR GRAFTS
Table 1: Marketed 6 mm ID Early CannulationI Vascular Grafts (qualified for use with the Adapter)
| Trade Name | Manufacturer | Catalog Number11 | Support Seal Required for HeRO Graft Adapter |
| FLIXENE® Standard Wall | Atrium Medical Corp. | 25053 | 25142 | 25052 | NO |
| GORE® ACUSEAL | W.L. Gore & Associates | ECH060010A | ECH060020A | ECH060040A ECH060050A. | NO |
APPROVED STANDARD WALL* VASCULAR GRAFTS
Table 2: Marketed 6 mm ID Standard WallI Vascular Grafts (qualified for use with the Adapter and Support Seal)
| Trade Name | Manufacturer | Catalog Number11 | Support Seal Required for HeRO Graft Adapter |
| IMPRA® | C.R. Bard | 05S06 | 10S06 | 20S06 | 30S06 | 40S06 | 50S06 60S06 | 70S06 | 80S06 | 90S06 | YES |
| GORE-TEX® | W.L. Gore & Associates | V06010L | V06020L | V06030L | V06040L V06050L | V06070L | V06080L | YES |
| GORE-TEX® Stretch | W.L. Gore & Associates | S0601 | S0602 | S0603 | S0604 | S0605 S0607 | S0608 | S0609 | YES |
| GORE® PROPATEN® | W.L. Gore & Associates | H060010A | H060040A | H060060A | H060080A | YES |
I. Refer to graft manufacturer Instructions for Use or website for indications and further information.
II. Catalog numbers may contain identifiers that are not reflected on this table. Consult the graft manufacturer’s website to determine which equivalent catalog numbers are available in your region.
* The Adapter should NOT be connected to any graft other than a new graft listed in Tables 1 and 2.